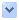Introduction to the Rotifera
Rotifers : the "wheel animalcules"
Rotifers are microscopic aquatic animals of the phylum Rotifera. Rotifers can be found in many freshwater environments and in moist soil, where they inhabit the thin films of water that are formed around soil particles. The habitat of rotifers may include still water environments, such as lake bottoms, as well as flowing water environments, such as rivers or streams. Rotifers are also commonly found on mosses and lichens growing on tree trunks and rocks, in rain gutters and puddles, in soil or leaf litter, on mushrooms growing near dead trees, in tanks of sewage treatment plants, and even on freshwater crustaceans and aquatic insect larvae. (Örstan, 1999)
Because of their very small size and mostly soft bodies, rotifers are not commonly favored for fossilization. Their only hard parts, their jaws, might be preserved in the fossil record, but their tiny size makes detection a serious challenge (Örstan, 1999). However, fossils of the species Habrotrocha angusticollis have been found in 6000 year old Pleistocene peat deposits of Ontario, Canada (Warner et al., 1988). The oldest reported fossil rotifers have been found in Dominican amber dating to the Eocene (Waggoner & Poinar, 1993).
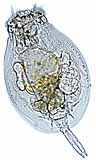
Rotifers : The rotifers are microscopic animals, and under high magnification will look something like the picture at upper left, for most perople using a light microscope. Those with more sophisticated microscopes and lighting techniques can give rotifers such as Philodina, grazing at lower left, a beautiful glow. At right, Collotheca is another monogonont rotifer, shown here bearing an egg on its stalk end. Notice the extemely long coronal cilia this rotifer uses to catch food. (Click on any of the pictures above for a larger image).
Rotifers are multicellular animals with body cavities that are partially lined by mesoderm. These organisms have specialized organ systems and a complete digestive tract that includes both a mouth and anus. Since these characteristics are all uniquely animal characteristics, rotifers are recognized as animals, even though they are microscopic. Most species of rotifers are about 200 to 500 micrometers long. However a few species, such as Rotaria neptunia may be longer than a millimeter (Orstan 1999). Rotifers are thus multicellular creatures who make make their living at the scale of unicellular protists.
 |
The name "rotifer" is derived from the Latin word meaning "wheel-bearer"; this makes reference to the crown of cilia around the mouth of the rotifer. The rapid movement of the cilia in some species makes them appear to whirl like a wheel.
At left, you can see a photomicrograph identifying basic anatomical features of Epiphanes brachionus. The general body plan of a rotifer consists of four basic regions: head, neck, trunk (body), and the foot. In most species, the head carries a corona (crown) of cilia that draws a vortex of water into the mouth, which the rotifer sifts for food. The food itself is ground by the trophi (jaws), located just behind the mouth in the pharynx (throat). Trophi are found in almost all rotifers, and are characteristic organs of the phylum Rotifera. The body of the rotifer is externally but not internally segmented. The body is telescopic, with a semi-flexible, extendible, transparent cuticle covering. It is the cuticle that suggests rotifers are close relatives of roundworms and arthropods. Within the body are the stomach and reproductive organs. The final region of the rotifer body is the foot; this foot ends in a "toe" containing a cement gland with which the rotifer may attach itself to objects in the water and sift food at its leisure. |
As rotifers are microscopic animals, their diet must consist of matter small enough to fit through their tiny mouths during filter feeding. Rotifers are primarily omnivorous, but some species have been known to be cannibalistic. The diet of rotifers most commonly consists of dead or decomposing organic materials, as well as unicellular algae and other phytoplankton that are primary producers in aquatic communities. Such feeding habits make some rotifers primary consumers. Rotifers are in turn prey to carnivorous secondary consumers, including shrimp and crabs.
As well as their morphology and feeding habits, reproduction in rotifers is rather unusual. Several types of reproduction have been observed in rotifers. Some species consist only of females that producetheir daughters from unfertilized eggs, a type of reproduction called parthenogenesis. In other words, these parthenogenic species can develop from an unfertilized egg, asexually. Other species produce two kinds of eggs that develop by parthenogenesis: one kind forms females and the other kind develops into degenerate males that cannot even feed themselves (sexual dimorphism). These individuals copulate resulting in a fertilized egg developing within the rotifer. The males survive long enough to produce sperm that fertilize eggs, which then form resistant zygotes that can survive if the local water supply should dry up. The eggs are released and hatch in the water. If the egg develops in the summer, the egg may remain attached to the posterior end of the rotifer until hatching.
A particular class of rotifers called bdelloids can be found living in almost all freshwater environments, and occasionally in brackish and marine waters. Bdelloids are known for their remarkable ability to survive drying through a process known as cryptobiosis. Factors determining the duration of time that a rotifer is able to withstand desiccation include the humidity and temperature at which they are kept. Ideally, more humid conditions and mild to warm temperatures prevent the very dry conditions that are unfavorable to rotifers. Rotifer eggs can also withstand drying, with older embryos having a greater chance at survival (Örstan, 1999). The species Brachonius calyciflorus has been found to conserve energy when food is scarce by decreasing its respiration rate, while other species show no change in respiration rate. It is predicted that the ability of some rotifer species to adapt to resources with temporal variation in availability allows the coexistence of competing species of rotifers. Therefore, there is a tradeoff between the competitive ability of rotifers and the maximum population growth rate for a particular species. (Kirk, 1999).
 Based on certain morphological similarities, rotifers and acathocephalans (the parasitic worms constituting the phylum Acanthocephala) have long been considered close relatives. Recent comparisons of 18S rRNA gene sequences provide further evidence of close relationship between these two groups. Rotifers and acanthocephalans have traditionally been classified as pseudocoelomates, along with a variety of other small worm-like animals. More recently, phylogenetic analyses have contradicted the hypothesis that Pseudocoelomata is a natural group. Instead, some pseudocoelomate animals, such as priapulids and nematodes, appear to be more closely related to arthropods, in a group termed Ecdysozoa. Other animals with a pseudocoel, such as rotifers and acanthocephalans, appear to be more closely related to Lophotrochozoa, a large alliance of protostomes that includes molluscs, annelids, brachiopods, etc.
Based on certain morphological similarities, rotifers and acathocephalans (the parasitic worms constituting the phylum Acanthocephala) have long been considered close relatives. Recent comparisons of 18S rRNA gene sequences provide further evidence of close relationship between these two groups. Rotifers and acanthocephalans have traditionally been classified as pseudocoelomates, along with a variety of other small worm-like animals. More recently, phylogenetic analyses have contradicted the hypothesis that Pseudocoelomata is a natural group. Instead, some pseudocoelomate animals, such as priapulids and nematodes, appear to be more closely related to arthropods, in a group termed Ecdysozoa. Other animals with a pseudocoel, such as rotifers and acanthocephalans, appear to be more closely related to Lophotrochozoa, a large alliance of protostomes that includes molluscs, annelids, brachiopods, etc.
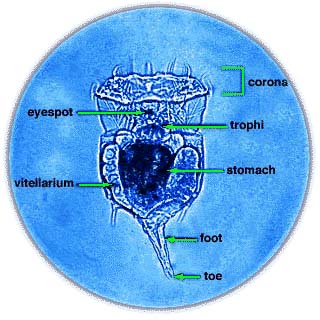
Phylum Rotifera is divided into three classes: Monogononta, Bdelloidea, and Seisonidea. The largest group is the Monogononta, with about 1500 species, followed by the Bdelloidea, with about 350 species. There are only two known species of Seisonidea, which is usually regarded as the most "primitive", and in morphological analyses it comes out in a basal position (see cladogram at right).
Observing rotifers is relatively uncomplicated with the correct procedure and equipment. When extracting rotifers from a sample, it is best to use a pipette, drawing water from the area around clumps of soil or plant matter in the sample. The sample should be quickly transferred to a slide so the rotifers do not adhere to the sides of the pipette. Additionally, cover-slips should not be used under a light microscope because rotifers are easily disturbed and may contract into an indiscernible ball. If food is added to the slide, rotifers can be observed swimming if they do not become stuck to the slide (Ricci, 1999). Thus, although rotifers are invisible to the naked eye, they can easily be watched in their exported natural environments with the help of a microscope.
For more information about Rotifera:
 Visit Microscopy UK for a wealth of wonderful rotifer images and information, including:
Visit Microscopy UK for a wealth of wonderful rotifer images and information, including:
Aydin Örstan has collected a wealth of bdelloid rotifer information for the web:
Additional sites rich in rotifer-nalia:
- International Association of Meiobenthologists, an organization for scientists who study the really tiny animals like rotifers.
- Laboratory Exercise on Bdelloid Rotifer Anatomy by Richard Fox. A step-by-step tecaching exercise involving rotifers.
- Population Dynamics study by James N. McNair, using rotifers in a chemostat.
- Rotifer Gallery by Ron Neumeyer, a talented photomicroscopist who has taken some very nice rotifer portraits.
- Rotifers Gallery, this time from Jerry Evans' information about Central Texas Natural History.
- Rotifer Study Methods, a wealth of free information on the collection, micromanipulation, and study of rotifers by Howard L. Taylor.
- Rotifer Systematic Database, coordinated by Elizabeth Walsh of the University of Texas at El Paso. The site includes morphological and molecular phylogenies, a database, references, and basic information.

Image of
Philodina kindly provided by Ron Neumeyer. Image of
Epiphanes brachionus internal anatomy prepared by Elizabeth Walsh, of the University of Texas at El Paso, and used with permission. Pictures of
Collotheca and
Lecane kindly supplied by Wim van Egmond from the "Gallery of Rotifers" listed above. Image of living rotifer by Molly McCarthy of Ohio University.
Sources:
- A. Jawahar Ali, S. S. S. Sarma, & H. J. Dumont, 1999. Cyst production in the fairy shrimp, Streptocephalus proboscideus (Anostraca) in relation to algal and loricated rotifer diet. Crustaceana (Leiden) 72(5): 517-530.
- Garey, J. R., Schmidt-Rhaesa, A., Near, T. J., Nadler, S. A. 1998. The evolutionary relationships of rotifers and acanthocephalans. Hydrobiologia 387-388: 83-91.
- Mary Jo Hartman & Stephen Sulkin, 1999. Effects of prior exposure to petroleum hydrocarbon contamination during brooding on the subsequent larval development of the brachyuran crab Hemigrapsus oregonensis. Journal of Crustacean Biology 19(4): 690-698.
- Kevin L. Kirk, et al., 1999. Physiological responses to variable environments: Storage and respiration in starving rotifers. Freshwater Biology 42 637-644.
- Thomas Nogrady, Robert L. Wallace, & Terry W. Snell. 1993. Rotifera, Vol. 1: Biology, Ecology and Systematics. The Hague: SPB Academic Publishing.
- Aydin Örstan, 1999. An Introduction to Bdelloid Rotifers. member.aol.com/bdelloid1/deloid.htm
- Claudia Ricci & Guilio Melone, 2000. Key to the identification of the genera of bdelloid rotifers. Hydrobiologia 418: 73-80.
- B. M. Waggoner & G. O. Poinar, Jr., 1993. Fossil habrotrochid rotifers in Dominican amber. Experientia (Basel) 49(4): 354-357.
- B. G. Warner, et al., 1988. Holocene fossil Habrotrocha angusticollis (Bdelloidea: Rotifera) in North America. Journal of Paleolimnology 1(2): 141-147.
- Wirz, A., Pucciarelli, S., Miceli, C., Tongiorgi, P., Balsamo, M. 1999. Novelty in phylogeny of Gastrotricha: Evidence from 18S rRNA gene. Molecular Phylogenetics and Evolution 13(2): 314-318.
- Xi Yilong & Huang Xiangfei. Effect of food supply in both food quality and quantity on the population dynamics of Brachionus urceolaris. Acta Hydrobiologica Sinica 23(3): 227-234.
--------------------------------------
Rotifer
From Wikipedia, the free encyclopedia
The rotifers, or rotifaers, make up a phylum of microscopic and near-microscopic pseudocoelomate animals. They were first described by Rev. John Harris in 1696 and other forms were described by Anton van Leeuwenhoek in 1703.[1] Most rotifers are around 0.1-0.5 mm long (although their size can range from 50μm to over 2 millimeters) [2], and are common in freshwater environments throughout the world with a few saltwater species. Some rotifers are free swimming and truly planktonic, others move by inchworming along the substrate, and some are sessile, living inside tubes or gelatinous holdfasts that are attached to a substrate. About 25 species are colonial (e.g., Sinantherina semibullata), either sessile or planktonic. Rotifers play an important part of the freshwater zooplankton, being a major foodsource and with many species also contributing to decompositioning of soil. [3]
[edit] Taxonomy and naming
The rotifera were first described by the Rev. John Harris in 1696, when he described a Bdelloid Rotifer as "an animal like a large maggot which could contract itself into a spherical figure and then stretch itself out again; the end of its tail appeared with a forceps like that of an earwig".[1] In 1702, Anton van Leeuwenhoek gave a detailed description of Rotifaer vulgaris and subsequently described Melicerta ringens and other species.[4] He was also the first to publish observations of the revivication of certain species after drying. Other forms were described by other observers, but it wasn't until the publication of Christian Gottfried Ehrenberg's Die Infusionsthierchen als vollkommene Organismen in 1838 that the rotifera were recognized as being multicellular animals.[4]
About 2200 species of rotifers have been described. Taxonomically, they are placed in the phylum Rotifera. This phylum is subdivided into three classes, Monogononta, Bdelloidea, and Seisonidea. The largest group is the Monogononta, with about 1500 species, followed by the Bdelloidea, with about 350 species. There are only two known species of Seisonidea.[5] The Acanthocephala, previously considered to be a separate phylum, has been unequivocally demonstrated to be modified rotifers. However, the exact relationship to other members of the phylum has not yet been resolved.[6]
[edit] Structure and form
Rotifers get their name (derived from Latin and meaning "wheel-bearer";[7] they have also been called wheel animalcules) from the corona, which is composed of several ciliated tufts around the mouth that in motion resemble a wheel. These create a current that sweeps food into the mouth, where it is chewed up by a characteristic pharynx (called the mastax) containing a tiny, calcified, jaw-like structure called the trophi. The cilia also pull the animal, when unattached, through the water. Most free-living forms have pairs of posterior toes to anchor themselves while feeding. Rotifaers have bilateral symmetry and a variety of different shapes. There is a well-developed cuticle which may be thick and rigid, giving the animal a box-like shape, or flexible, giving the animal a worm-like shape; such rotifers are respectively called loricate and illoricate.
Like many other microscopic animals, adult rotifaers frequently exhibit eutely - they have a fixed number of cells within a species, usually on the order of one thousand.
Males in the class Monogononta may be either present or absent depending on the species and environmental conditions. In the absence of males, reproduction is by parthenogenesis and results in clonal offspring that are genetically identical to the parent. Individuals of some species form two distinct types of parthenogenetic eggs; one type develops into a normal parthenogenetic female, while the other occurs in response to a changed environment and develops into a degenerate male that lacks a digestive system, but does have a complete male reproductive system that is used to inseminate females thereby producing fertilized 'resting eggs'. Resting eggs develop into zygotes that are able to survive extreme environmental conditions such as may occur during winter or when the pond dries up. These eggs resume development and produce a new female generation when conditions improve again. The life span of monogonont females varies from a couple of days to about three weeks.

Scanning electron micrographs showing morphological variation of bdelloid rotifers and their jaws.
Bdelloid rotifers are unable to produce resting eggs, but many can survive prolonged periods of adverse conditions after desiccation. This facility is termed anhydrobiosis, and organisms with these capabilities are termed anhydrobionts. Under drought conditions, bdelloid rotifers contract into an inert form and lose almost all body water; when rehydrated, however, they resume activity within a few hours. Bdelloids can survive the dry state for prolonged periods, with the longest well-documented dormancy being nine years. While in other anhydrobionts, such as the brine shrimp, this desiccation tolerance is thought to be linked to the production of trehalose, a non-reducing disaccharide (sugar), bdelloids apparently lack the ability to synthesise trehalose.
Bdelloid rotifer genomes contain two or more divergent copies of each gene, suggesting a long term asexual evolutionary history.[8] Four copies of hsp82 are, for example, found. Each is different and found on a different chromosome excluding the possibility of homozygous sexual reproduction.
[edit] Feeding
Rotifiers eat fish waste, dead bacteria and algae. They eat particles up to 10 micrometre in size. Rotifiers filter water at the rate of 100,000 times its own volume per hour. They are used in fish tanks to help clean the water, to prevent clouds of waste matter.
[edit] References
- ^ a b Harmer, Sidney Frederic and Shipley, Arthur Everett (1896). The Cambridge Natural History. The Macmillan company. pp. 197. http://books.google.com/books?id=bvoEAAAAYAAJ&pg=PA197&lpg=PA197&dq=%22john+harris%22+rotifer&source=web&ots=hhpqBzp2L4&sig=XXsKKHav5eiRZVu104NbDQvtvwI&hl=en&sa=X&oi=book_result&resnum=3&ct=result. Retrieved on 2008-07-25.
- ^ [1] Welcome to the Wonderfully Weird World of Rotifers by Richard L. Howey
- ^ [[2]]
- ^ a b Bourne, A.G. (1907). Baynes, Spencer and W. Robertson Smith. ed. Encyclopaedia Britannica. XXI (Ninth Edition ed.). Akron, Ohio: The Werner Company. pp. 8. http://books.google.com/books?id=yAsEAAAAYAAJ&printsec=titlepage#PPA8,M1. Retrieved on 2008-07-27.
- ^ Baqai, Aisha; Guruswamy, Vivek; Liu, Janie; and Rizki, Gizem (2000-05-01). "Introduction to the Rotifera". University of California Museum of Paleontology. http://www.ucmp.berkeley.edu/phyla/rotifera/rotifera.html. Retrieved on 2008-07-27.
- ^ Shimek, Ronald Ph.D (January 2006). "Nano-Animals, Part I: Rotifaers". Reefkeeping.com. http://reefkeeping.com/issues/2006-01/rs/index.php. Retrieved on 2008-07-27.
- ^ Pechenik, Jan A. (2005). Biology of the invertebrates. Boston: McGraw-Hill, Higher Education. pp. 178. ISBN 0072348992.
- ^ J.L.M. Welch, D.B.M Welch, and M. Meselson. Cytogenic evidence for asexual evolution of bdelloid rotifers. Proc. Nat. Acad. Sci., Feb. 2004 vol. 101, no. 6, pp.1618-1621
[edit] External links
輪形動物
出典: フリー百科事典『ウィキペディア(Wikipedia)』
輪形動物は、いわゆるワムシ類と総称される動物の分類群である。
概説 [編集]
輪形動物門 (Rotifera) は、ワムシと呼ばれる水中の微小動物からなる動物群である。主として淡水に生息し、若干の海産種や陸生種がある。多くは1 mm に満たず、たいていは100-500 μm 程度の大きさである。浮遊生活か、藻類や沈殿物の表面を匍匐して暮らしている。一部に固着性の種がある。世界で約3,000種が知られる。
単為生殖をする種が多く、雄が常時出現する例は少ない。雄が全く見られない群もある。なお、雄は雌よりはるかに小さく、形態も単純で消化管等も持たない。以下の構造等の記述は主として雌に関するものである。
外部形態 [編集]

輪形動物の
走査型電子顕微鏡写真。
(A)
Rotaria neptunia(B)
R. macrura(C)
R. tardigrada(D)
R. sordida(E)
R. tardigrada(口器のみ)
壷型の胴体と、後方に伸びる尾部を持ち、頭にある繊毛を使って運動する。全体としては左右相称で、腹背の区別はあるが、さほどはっきりしない例もある。
体の先端部は幅広く、ここには繊毛が円をなして配置し、繊毛冠(Corona)を形成する。この繊毛は摂食にも運動にも使われる。この部分は形態的にははっきりしない場合もあるが、頭部と言われる。眼点や特殊な感覚器を備える例もある。
頭部に続く部分は胴部で、円筒形から壷型、内臓の大部分がここに収まる。体表はキチン質の表皮に覆われる。がっしりとした被甲に覆われる例も多い。
それに続く尾部は、いくつかの節に分かれて、よく伸縮する。先端に二本の指と爪があり、また粘液腺などを持って体を支えるのに使われる。匍匐性の種では胴と同じくらいの幅と長さを持ち、節があるものもあるが、体節とは認められていない。また、この付近に卵をぶら下げて活動するものがよくある。
多くのものは付属肢を持たないが、ミジンコワムシは二対の付属肢があり、それを使って泳ぐ。また、可動の棘を持つものもある。ミツウデワムシは胴部前方に一対、後方に一本の棘があり、この前方の一対を大きく動かして撥ねるように泳ぐ。
内部構造 [編集]
消化系は直線的。繊毛冠の中央に口が開き、胴部の前端付近に咽頭部がある。この部分は厚い筋肉に覆われ、石灰質の咀嚼板が組合わさって咀嚼器を構成している。 それに続いて胃と腸があり、肛門は胴部の後端にある。フクロワムシは腸と肛門を欠く。
胴部の内部を広く占める体腔内は、消化系の表面に上皮層を欠くので偽体腔である。また、縦に走る筋肉がよく発達し、これによって体を伸び縮みさせ、よく運動する。
神経系としては咀嚼嚢の背面に脳神経節があり、ここから全身に末梢神経が走る。 排出系は原腎管を左右一対持ち、その末端は肛門につながる膀胱に開く。生殖巣は消化器の腹側にあり、やはり肛門に口を開く。なお、雄ではこの位置に陰茎がある。
生態 [編集]
基本的には水中動物であり、陸で見られるものも、特に湿った状態の時に出現する。繊毛を動かしてデトリタスなどを集めて食べているものが多いが、植物の汁を吸うもの、捕食性で原生動物や他のワムシ類などを捕らえるものも知られる。寄生性のものも知られている。
多くは自由生活で、浮遊性のものもあれば、基質上をはい回ることの多いものもある。繊毛を動かして泳ぐか、尾部で基質表面に付着し、尾を動かして運動する。ヒルガタワムシは頭部と尾部を使い、ヒルやシャクトリムシのように這う。
固着性の種もあり、それらは基質表面に棲管を作り、そこに体をいれ、伸び出して管の口から繊毛冠を広げる。その仲間で変わっているのはテマリワムシで、多数個体が互いに尾の先端でくっつき合い、それが寒天質に包まれてくす玉のような群体となり、水中を回転しながらただよう。
生活環 [編集]
多くの種が単為生殖をする。それらは条件のいい間は夏卵と言われる殻の薄い卵を産み、この卵はすぐに孵化して雌となり、これを繰り返す。条件が悪化するなどの場合には減数分裂が行われて雄が生まれ、受精によって生じた卵は休眠卵となる。休眠卵は乾燥にも耐え、条件がよくなれば孵化する。なお、ヒルガタワムシ類では雄は全く知られていない。他方、ウミヒルガタワムシでは雄が常時存在することが知られる。
なお、単為生殖を繰り返す期間に、殻の角が伸びるなど形態が世代を繰り返す間に変化する例があり、周期的体型輪廻(Cyclomorphosis)と言われる。
利害 [編集]
特に害をなす局面はない。
野外ではさまざまな水域で生息密度も高く、小魚等の重要な餌になっている。また、デトリタス食のものは水の浄化にも効果が高いと言われる。特にシオミズツボワムシは人工培養も行われており、アルテミアよりも小さな生き餌として、養魚場等では重宝される。
系統 [編集]
偽体腔を持つとされ、かつて袋型動物とされたこともある。その中では小型であること、繊毛で運動する点で腹毛動物に似ている。原腎管を持つ点では扁形動物と共通点がある。
その体制をトロコフォアと対比させる説が唱えられたこともある。
分類 [編集]
すべてを輪型動物綱にまとめ、伝統的には生殖器によって三群に分けるが、あまり安定した体系はないようである。以下に旧来の分類体系を示す。
輪虫綱 Rotatoria
- ウミヒルガタワムシ綱 Seisonidea:首がある。寄生性。雌雄とも常在。
- ヒルガタワムシ綱 Bdelloidea(二生殖巣綱 Digononta):体は細長く、節があり、ヒルのように運動。雄は知られていない。
- ヒルガタワムシ目 Bdelloidea
- ヒルガタワムシ科 Philodidae:ヒルガタワムシ Rotaria・ヤドカリワムシ Mniobia・Dissotrocha・Philodina・Macrotrochela
- 単生殖巣目 Monogononta:卵巣は1。雄は退化的。
- ハナビワムシ目 Collothecaceae
- ハナビワムシ科 Collithecidae:ハナビワムシ Collotheca・ハナカザリワムシ Stephanoceros
- マルサヤワムシ目 Flosulariaceae
- ワムシ目(遊泳目) Ploima
- ミツウデワムシ科 Filiniidae:ミツウデワムシ Filinia
- ミジンコワムシ科:ミジンコワムシ Hexartha
- ヒラタワムシ科 Testudinellidae:ヒラタワムシ Testudinella・Pompholyx
- コガタワムシ科 Notommatidae:コガタワムシ Notommata・カシラワムシ Cephalodella
- ドロワムシ科 Synchaetidae:ドロワムシ Synchaeta・ハネウデワムシ Polyarthra
- スジワムシ科 Ploesomatidae:スジワムシ Ploesoma
- フタエワムシ科:フタエワムシ Chromogaster・トクリワムシ Gastropus
- ネズミワムシ科 Trichocercidae:ネズミワムシ Trichocerca
- フクロワムシ科 Asplanchnidae:フクロワムシ Asplanchna
- ツボワムシ科 Brachionidae:ツボワムシ Brachionus・オケワムシ Platyas・カメノコウワムシ Keratella・シマワムシ Notholca・トゲナガワムシ Kellicottia・ミズワムシ Epiphanes・ハオリワムシ Euchlanis・フタエワムシ Dipleuchlanis・ウサギワムシ Lepadella・ツノサヤワムシ Mytilina・オニワムシ Trichotria・オオトゲワムシ Macrochaetus・チビワムシ Colurella・ツキガタワムシ Lecane
参考文献 [編集]
- 白山義久編集;岩槻邦男・馬渡峻輔監修『無脊椎動物の多様性と系統』(2000)裳華房
- 水野 彦『日本淡水プランクトン図鑑』(1964)保育社
- 椎野季雄,『水産無脊椎動物学』,1969,培風館
- 岡田要,『新日本動物図鑑』,1976,図鑑の北隆館
---------------------
輪形動物門 輪虫綱
** ワムシに属する生物 **
| ◎・・極めて普通に見られる |
○・・やや普通に見られる |
| △・・あまり普通でない |
×・・極めて珍しい |
このページのトップへ
** ワムシの特徴 **
- 頭部に輪状に繊毛が生じています(図1の赤矢印)。この繊毛を激しく動かして泳ぎます。
- 種類により形はさまざまです。
- 多細胞動物で,体内に内臓等の諸器官があります。
- 種類によっては体の後部に足(図2の赤矢印),およびその先に趾(あしゆび:図2の青矢印)とよばれる2またに分かれた部分があります。(注1も見てください)
- 種類によっては体をおおう硬い被甲(図2の緑矢印)があります。(注2も見てください)
- 小形~中形で,肉眼で見えるものはほとんどありません。
注1:足と趾がはっきりわかるもの,足および趾が全くないもの,足が短くて趾だけがあるように見えるものなどいろいろあります。この足,趾の有無や形がワムシの分類には大変重要になる場合が多いです。
注2:被甲がなく変形するもの,被甲がやわらかいもの,被甲が硬いものなどいろいろあります。この被甲の有無や形がワムシの分類には大変重要になる場合が多いです。
このページのトップへ
** 似ているものと見分け方 **
- 繊毛虫 : ワムシも繊毛虫もともに種類数が多く,中には互いに形が似ているものがあります。また,ともに少なくとも体の一部に繊毛をもっています。しかし,繊毛虫は単細胞生物で,体内に内臓等の諸器官はありませんが,ワムシは多細胞動物で,体内に内臓等の諸器官があります。
- イタチムシ : ワムシとイタチムシは分類学上近い位置にあるだけに,形が似ている場合がありますが,イタチムシの体の形,特に頭部の形は独特のもので,これと見分けがつかないほど似た形のワムシはありません。また,ワムシは,イタチムシのように全身に毛が生えていたり,全身がうろこでおおわれていたりすることはありません。
このページのトップへ
** 注意 **
- 輪形動物門(りんけいどうぶつもん)には,イタチムシを含めることも多いです。また,ワムシは,イタチムシやセンチュウなどいっしょに袋形動物門(たいけいどうぶつもん)というグループに入れられる場合があります。
- ワムシでは,属を代表する種に属と同じ和名がつけられているものが数多くあります。ヒラタワムシ,フクロワムシ,ツボワムシなど,例はたくさんあります。ここでは,単に「○○ワムシ」といった場合にはその属全体(たとえば,「ツボワムシ」なら,ツボワムシ属(Brachionus)全体)をさします。属名と同じ和名の種のことを表すときは,たとえば「ツボワムシ(Brachionus calyciflorus)」のように,種の学名を併記した形で表記します。
- ワムシには体がやわらかく伸び縮みするもの,被甲に突起があるもの,長い足や趾をもつものなどがあり,どこまでを体長とするか迷うものが多いです。ここでのワムシの体長は,被甲がなく体が伸び縮みするものは,写真に写った状態での長さ,被甲を持つものは突起を除いた被甲の長さとします。
- ワムシは種類が多く,いろいろな形のものがいます。ここにのせていないもので,形が違っているものもいます。
- ワムシは,オスとメスで形が全く違います。しかし,オスは極めてまれで,研究も進んでおらず,専門知識をもった人でなければワムシのオスを見定めることは無理といってよいでしょう。ここにのせた写真は全部メスです。
- ワムシは見る方向によって形が全く違って見えるのが普通です。
- しばしば体の後部に楕円形のもの(図3の赤矢印)をくっつけているものが見られますが,これは卵です。また,図4のように卵だけが見られる場合もあり,初心者には判断が難しい場合があります。また,図5のように孵化する寸前の卵は,ワムシだとはわかっても,何ワムシなのかわからないということになる可能性があります。ワムシの卵を参照してください。
 |
 |
 |
 |
 |
| 図3:卵を持ったワムシ(ツボワムシ) |
|
図4:ワムシの卵 |
|
図5:孵化する寸前のワムシの卵 |
このページのトップへ
トップページ|名前を調べる
顯示文章地圖
文章定位: